Electronic Structure
Each electron in an atom can be described by four quantum numbers: n, l, m, and s.
n, the principal quantum number, describes the size of the orbital.
l describes the shape, e.g. s signifies a spherical shape, p polar, d cloverleaf.
m describes the orientation, e.g. an s orbital can be oriented in only one way, a p orbital can be oriented along the x axis, y axis, or z axis.
s describes the spin of an electron, which is either clockwise or counterclockwise (+½ or -½).
- Aufbau principle
- Atomic orbitals are filled one at a time, starting with the orbital that has the lowest energy.
Hund's rule
- Electrons added to degenerate orbitals (i.e. that have the same energy, e.g. 2p) have parallel spins until each of the orbitals has one electron before a second electron is placed in one of these orbitals.
Pauli exclusion principle
- No two electrons in an atom can have the same set of values for all four quantum numbers n, l, m, and s.
| Principal quantum number n | Shape l | Orientation m | Spin s | Number of electrons needed to fill subshell |
1 | s | 1 orientation: | ± ½ | 2 |
2 | s | 1 orientation: | ± ½ | 2 |
2 | p | 3 orientations: | ± ½ | 6 |
3 | s | 1 orientation: 0 | ± ½ | 2 |
3 | p | 3 orientations: | ± ½ | 6 |
3 | d | 5 orientations | ± ½ | 10 |
4 | s | 1 orientation: | ± ½ | 2 |
4 | p | 3 orientations: 4Px, 4Py, 4Pz | ± ½ | 6 |
4 | d | 5 orientations | ± ½ | 10 |
4 | f | 7 orientations | ± ½ | 14 |
Etc. |
-
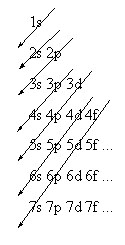
To determine the sequence in which orbitals are filled, follow the arrows in the diagram below.